Brain Tumors
Glioblastoma (GBM) is the most common and aggressive form of primary brain tumor. It is the highest grade (WHO grade IV) of astrocytoma. The current standard-of-care treatment involves surgical resection followed by radiation and chemotherapy with temozolomide (TMZ). However, despite providing best of the currently available treatments the median survival of GBM patients remains only ~15-17 months post diagnosis. This highlights the importance of new research to find out alternative avenues to treat GBM.
The reasons why these tumors are so difficult to treat lie within the inherent properties of these tumor cells. GBM cells are highly invasive in nature. They show extensive single cell infiltrations and long distance migrations in the brain. While it is possible to remove the large tumor mass by surgical resection, it is impossible to detect or remove the small tumor masses or single cell infiltrates that are indistinguishable from the healthy tissues around. These residual tumors later give rise to recurrence. Recurrent tumors are also often resistant to radio- or chemotherapy, ultimately causing patients’ demise.
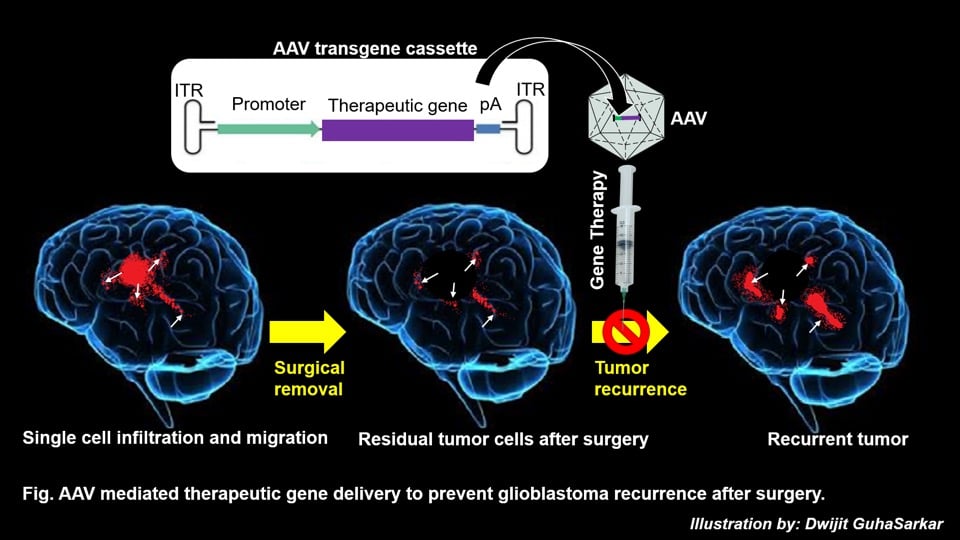
We believe that gene therapy has the potential to prevent this recurrence of tumor and thereby can improve the survival of patients. AAV vectors have currently emerged to be one of the safest and most attractive gene delivery vehicles for the treatment of CNS disorders. We have come up with a new paradigm of AAV mediated gene therapy approach to treat GBM tumors. We have demonstrated that by delivering genes of secretory therapeutic proteins to healthy brain cells we can create a microenvironment in the brain that is non-permissive to tumor growth (Maguire et al., 2008). The advantage of using secretory therapeutic molecule is that a relatively small number of transduced cells can function as the therapeutic protein factories and the secreted protein molecules can therapeutically impact a larger number of neighboring tumor cells (bystander effect). Using both xenograft and syngeneic mouse models we have shown that local intracranial delivery of AAVs encoding the therapeutic gene can effectively treat established aggressive human and mouse glioblastoma. We have also shown that combination of this AAV gene therapy approach with standard chemotherapeutic drug TMZ prolongs the survival of animals significantly over any single mode of treatment. Using AAV9 vectors, that can transduce the brain cells efficiently upon systemic delivery, we have further demonstrated that intravenous injection of AAVs carrying therapeutic gene can completely eliminate orthotopically established highly invasive human GBM from a xenograft mouse model. Moreover, we have shown that systemic delivery is a superior route of gene delivery over local injection for treatment of distant multifocal invasive tumors (GuhaSarkar et al., 2016).
Also recently we are initiating studies directed at understanding the contribution of different genes to the migratory behavior of GBM cells and strategies to counteract it. In collaboration with other labs we are also exploring the possibilities of using other modes of gene therapies or RNAi therapies for GBM.
