Welcome to the Torres Laboratory
Eduardo Torres Ph.D email
Associate Professor
Department of Molecular, Cell and Cancer Biology (faculty profile)
Our research aims to determine the molecular mechanisms underlying the effects of aneuploidy (an abnormal number of chromosomes) on cellular physiology. The motivation to understand how cells respond to aneuploidy comes from the fact that cancer cells almost always harbor chromosome numbers higher than the normal count of 46. Despite aneuploidy being a hallmark of cancer cells, its role in tumorigenesis remains poorly understood. Recently, our studies showed that acquiring one extra copy of any chromosome lowers cellular fitness and proliferation. However, despite being aneuploid, a hallmark of cancer cells is unlimited proliferation potential. Therefore, we hypothesize that cancer cells must evolve and adapt to overcome the negative consequences of aneuploidy to cellular fitness. See our publications where we discovered that mutations in genes that regulate protein stability (Dephoure et al. 2014) or cellular metabolism (Hwang et al. 2017, 2019) improve aneuploid cells' cellular fitness. A review of our work over recent years is now available in Chromosome Research (Torres E.M., 2023).
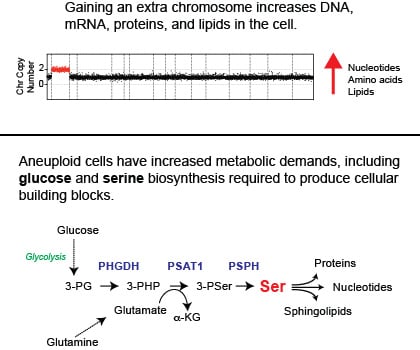
Studying the consequences of aneuploidy is relevant to human health because Down syndrome is the most common genetic disease in humans. Individuals born with an extra copy of chromosome 21 (47 chromosomes) have Down syndrome. Most research on Down syndrome has focused on identifying the mechanisms by which a third copy of a given gene on chromosome 21 causes the disease. We recently showed that cells with three copies of chromosome 21 have cellular defects associated with aneuploidy. These defects are present in human, mouse, or yeast cells that harbor an extra chromosome. Therefore, we hypothesize that in addition to the increased activity of genes on chromosome 21, the consequences of aneuploidy on cellular physiology are essential to understand better how an extra copy of 21 causes Down syndrome. See our latest publication on this topic (Hwang et al. 2021).
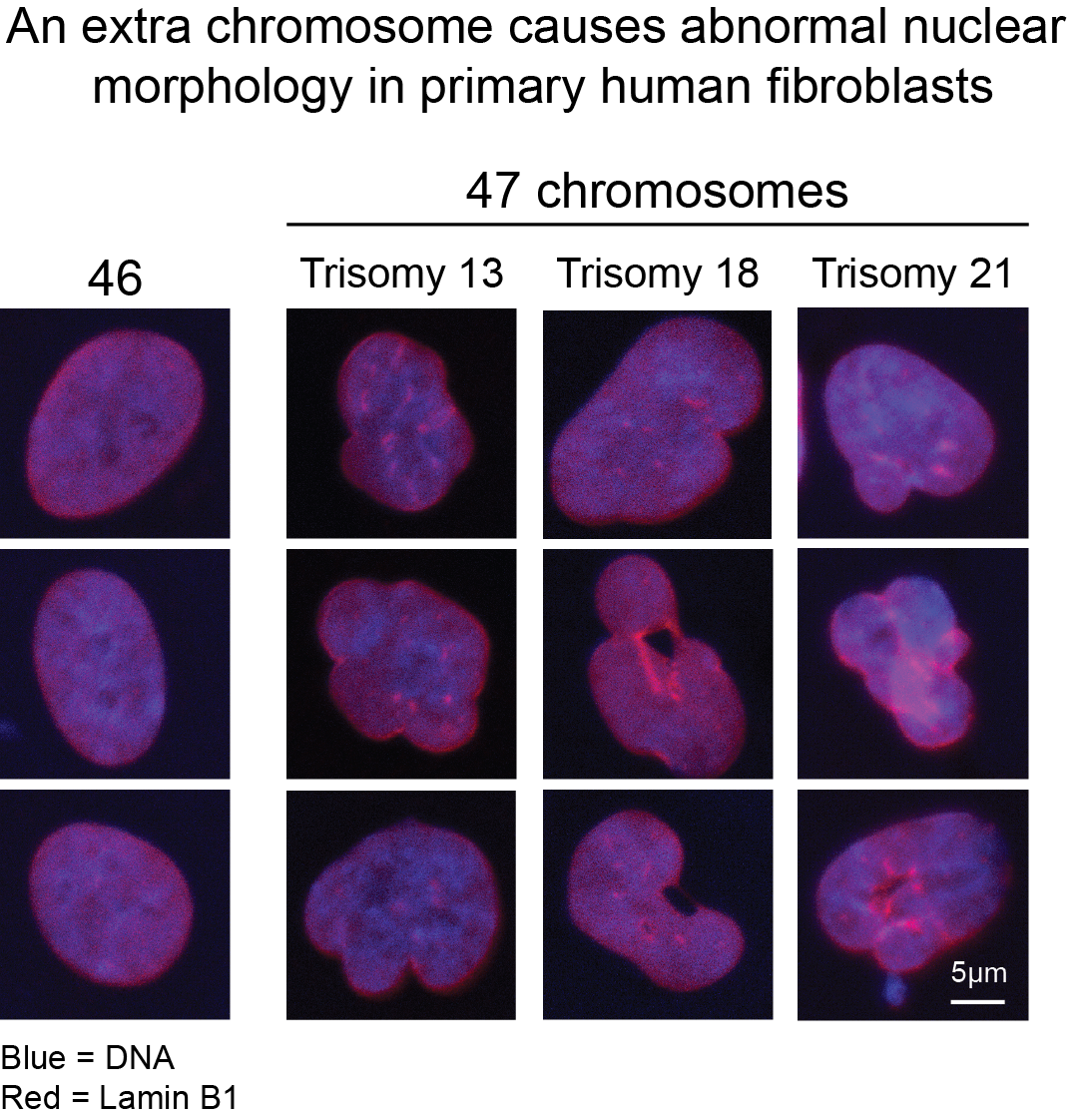
An abnormal nuclear morphology is a conserved phenotype of aneuploid cells in yeast and human cells. Several projects in the lab focus on understanding how extra chromosomes disrupt the morphology of the nucleus and determining the physiological consequences of this phenotype.
Interested in studying aneuploidy? WE ARE HIRING! e-mail eduardo.torres@umassmed.edu