UMass Radiology to participate in PRESERVE study
Date Posted: Tuesday, March 03, 2015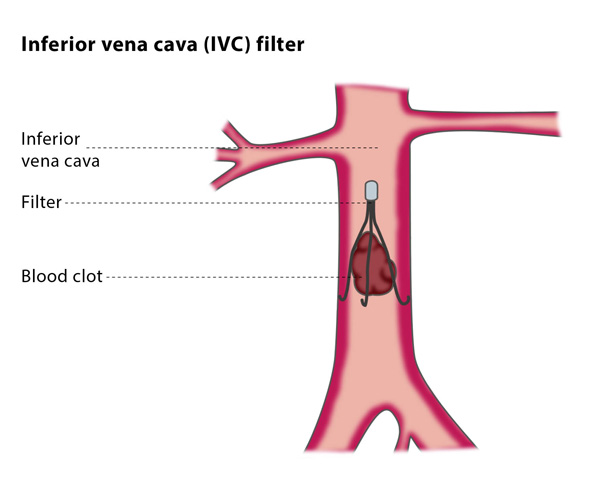 In August of 2010, the FDA sent out a medical alert warning Doctors and patients about the potential health risks of Inferior Vena Cava (IVC) filters. The FDA recommended that Doctors should consider removing the retrievable filters due to concerns of them moving or breaking. The Society of Interventional Radiology and The Society for Vascular Surgery have responded to this alert by launching “PRESERVE - the first large-scale, multispecialty prospective study to evaluate the use of inferior vena cava (IVC) filters and related follow-up treatment”. PRESERVE standing for PRE dicting the Safety and Effectiveness of InferioR VEna Cava Filters.
In August of 2010, the FDA sent out a medical alert warning Doctors and patients about the potential health risks of Inferior Vena Cava (IVC) filters. The FDA recommended that Doctors should consider removing the retrievable filters due to concerns of them moving or breaking. The Society of Interventional Radiology and The Society for Vascular Surgery have responded to this alert by launching “PRESERVE - the first large-scale, multispecialty prospective study to evaluate the use of inferior vena cava (IVC) filters and related follow-up treatment”. PRESERVE standing for PRE dicting the Safety and Effectiveness of InferioR VEna Cava Filters.
On February 28th of this year, the UMass Department of Radiology was selected to participate in this 5-year IVC filter study. The goal: to gain a comprehensive view of most of the IVC filters used in the United States. The Society of Interventional Radiology says, “The five-year study will evaluate the overall safety and efficacy of filters placed by doctors and intends to enroll 2,100 patients at approximately 60 centers in the United States. There will be at least 300 patients enrolled for each participating manufacturer filter, and patients will be evaluated every six months post procedure up to 24 months or filter retrieval.”
To learn more about this study visit:
http://www.prnewswire.com/news-releases/large-scale-study-on-vein-filter-use-launches-282455991.html
http://evtoday.com/pdfs/et1112_F7_Johnson.pdf
SOURCES:
“Medical Societies to Launch Large-Scale Study on Vein Filter Use”. Vascular Web. The Society of Interventional Radiology and Society for Vascular Surgery. 13 February 2015. Web. 1 March 2015.
“Large-scale study on vein filter use launches”. PR News Wire. The Society of Interventional Radiology and Society for Vascular Surgery. 12 Nov. 2014. Web. 1 March 2015.